Energy Efficient House
Questions:
How can we design a cold frame that is comfortable in all conditions without use of electricity?
What is the most efficient way to heat and cool a cold frame?
How can we get the light in through the Northern Window of the Solar Studio?
How can we design a cold frame that is comfortable in all conditions without use of electricity?
What is the most efficient way to heat and cool a cold frame?
How can we get the light in through the Northern Window of the Solar Studio?
Spencer, Eric, Ryan and I worked on this project for around two months. This project was the longest project we've ever done in STEM program. We had two goals. One, we had to design the solar reflector panels for the light to get into the Northern Window of the Solar Studio. Second, we had to design a cold frame for allowing plant growth during winter in San Marin Garden. This project contained 9 main steps.
1. Atomic structure, state, and heat
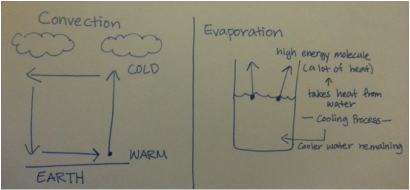
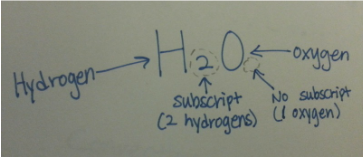
Some of the basic facts about atoms, heat transfer, and solids, liquids, gases. ✸Atom •Nucleus:center of Atom Proton (+) Neutron(∅) Electron(—) Examples of Atom- Helium, Carbon, Gold, etc (click: periodic table) •Molecule: multiple atoms bonded together •Compound: type of molecule with different types elements bonded Examples- H2O, CO2, C6H12O6, O2, ect (click: Periodic table) •Atoms are incredibly small and very plentiful Example- 45,000,000,000,000,000,000 atoms= sugar cube air •Isotope: an atom with unusual number of neutrons Example- C-14 •Ion: an atom with unusual number of electrons Example- Na+, H+, Cl− •Almost all mass in atom is in nucleus and nucleus is very small |
✸Heat Transfer •Almost all Earth's energy comes from the Sun. •Sun evergy travels 91million miles through space. •Conduction: transfer heat through solid Example- pan on stove •Convection: transfer heat through fluid(liquid/gas) Example- 1st floor & 2nd flower of a house •Radiation: transfer of heat through waves/particles/rays Example- Sunlight •Insulation: bad at conducting (opposite of conduction), resistance to transferring heat •Heat= form of energy (thermal energy) Unit: Joules ( J ). A measure of the Kinetic Energy of atoms •Thermal Conductivity: the rate at which a material transfers heat •Thermal Resistance: the ability of material to resist transferring heat |
✸Solid, Liquids, Gases
•Heat is a measure of the movement of molecules
More heat = More movement
•Solid: all molecules are held in place
•Liquid: all molecules are bonding with each other but they can change bonds to flow (more movement)
•Gas: all molecules don't/can't have to bond with each other. More movement=take up way more space and less dense.
2. Design a hot water heater
Our first part of our project was to design and build a hot water heater. We constructed a very simple water heater using a black aluminum box, whiter poster papers, a PVC pipe, and an aluminum foil. We started by cutting out 7 different isosceles triangles and cover each of them with aluminum foil. Then, we connected all 7 triangles to make a parabolic reflector. After that, we connected the black aluminum box with a PVS pipe and stuck it in the middle of our parabolic reflector. Lastly, we put water(720 g= 720mL) into the black aluminum box to begin the experiment.
How our device works:
•We used a 4 feet wide and 1 foot deep parabolic reflector
•The reflector reflected the radiation to one point (the middle: black aluminum box)
•Black painted aluminum box for insulation
•Reflector angles at 68° to create vertical radiation light (The stand helped to keep it in the angle)
Perpendicular to the sun's rays
We left the it out in the sun for 30 minutes. Our initial temperature of the water was 17°C. After 30 minutes, the temperature of the water raised to 47°C. We calculated the heat gained by the using the equation below:
Q= mCwΔT
Q= 720g (4.186J) (30°C)
Q= 90,417.6 J
Our group Heat gain: 90,417.6 J
Our first part of our project was to design and build a hot water heater. We constructed a very simple water heater using a black aluminum box, whiter poster papers, a PVC pipe, and an aluminum foil. We started by cutting out 7 different isosceles triangles and cover each of them with aluminum foil. Then, we connected all 7 triangles to make a parabolic reflector. After that, we connected the black aluminum box with a PVS pipe and stuck it in the middle of our parabolic reflector. Lastly, we put water(720 g= 720mL) into the black aluminum box to begin the experiment.
How our device works:
•We used a 4 feet wide and 1 foot deep parabolic reflector
•The reflector reflected the radiation to one point (the middle: black aluminum box)
•Black painted aluminum box for insulation
•Reflector angles at 68° to create vertical radiation light (The stand helped to keep it in the angle)
Perpendicular to the sun's rays
We left the it out in the sun for 30 minutes. Our initial temperature of the water was 17°C. After 30 minutes, the temperature of the water raised to 47°C. We calculated the heat gained by the using the equation below:
Q= mCwΔT
Q= 720g (4.186J) (30°C)
Q= 90,417.6 J
Our group Heat gain: 90,417.6 J
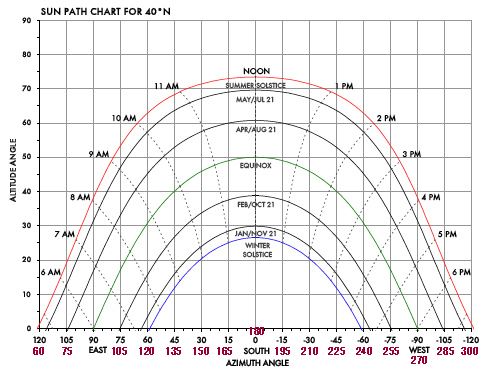
3. Solar angles and how it changes during seasons
✸Sun Angles
•Vertical Light: brightest and most concentrated, therefore there is more light and heat per unit area
•Slanted Light: more spread out, therefore less concentrated, less heat, and less brightness per unit area
•The hemisphere tilted towards the sun, get more vertical light- SUMMER
•The hemisphere tilted away from the sun, get more slanted light- WINTER
•The effects of the spherical Earth
The latitudes near the equator always get vertical light
The latitudes far from the equator always get slanted light
✸Sun Angles
•Vertical Light: brightest and most concentrated, therefore there is more light and heat per unit area
•Slanted Light: more spread out, therefore less concentrated, less heat, and less brightness per unit area
•The hemisphere tilted towards the sun, get more vertical light- SUMMER
•The hemisphere tilted away from the sun, get more slanted light- WINTER
•The effects of the spherical Earth
The latitudes near the equator always get vertical light
The latitudes far from the equator always get slanted light
Solar angles throughout the days, and during the year:
4. Day lighting design activity
For this part of our project, we learned about different day lighting techniques and features. From this, we built a small model house and added day lighting features such as solar tubes, light shelves, clerestory windows, and skylights. You can replace all the electric lights with following:
Solar Tubes: A reflective tube to bring daylight into homes.
Light Shelves: A window sill that reflects daylight onto the ceiling and the room
Clerestory Windows: A short, close to top of the wall window. Used to bring midday(slanted) light into the room.
Skylights: A window in a roof to let overhead daylight to come in
For this part of our project, we learned about different day lighting techniques and features. From this, we built a small model house and added day lighting features such as solar tubes, light shelves, clerestory windows, and skylights. You can replace all the electric lights with following:
Solar Tubes: A reflective tube to bring daylight into homes.
Light Shelves: A window sill that reflects daylight onto the ceiling and the room
Clerestory Windows: A short, close to top of the wall window. Used to bring midday(slanted) light into the room.
Skylights: A window in a roof to let overhead daylight to come in
|
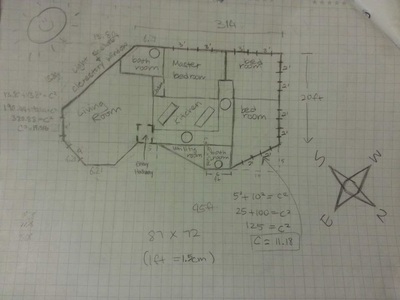
5. Site Selection
For this, we had to look for a perfect place where it will be pleasing in every factors for people to live in. We had to considerate the temperature, sun/shade, privacy, view, space, landscape, nondestructive, integrated into nature, accessibility, and visible to public. Considering all these factors, we decided on the southeast of San Marin Art classroom. |
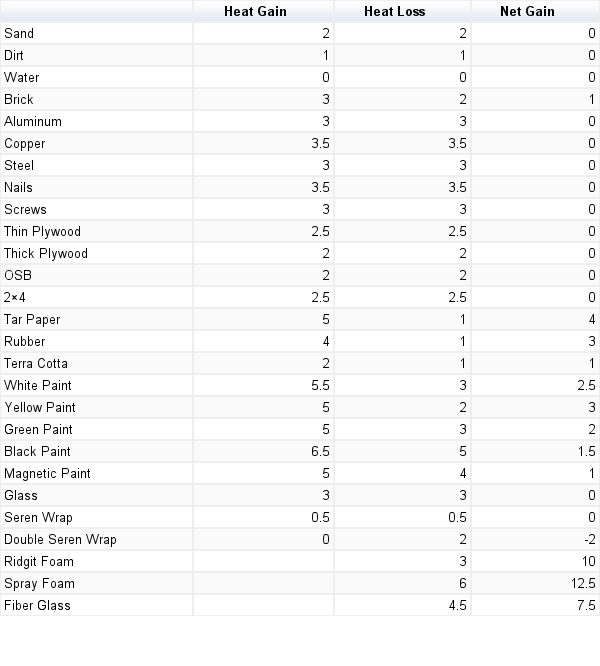
6. Materials Testing
For the first time, we made up a lab as a class. We had to design an experiment to test all of the possible materials we might use in the interior or exterior of our structure. First, we came up with a problem to solve. Which building materials are the most effective conductor/insulator/absorbing/sustaining heat? Then, we came up with materials list:
Conducting: Copper, plexiglas, aluminum, steel, plywood, OSB, screws, and nails
Insulating: double & single paned window, fiberglass insulation, magnifying glass, ridged foam, and spray foam.
Absorbing & Holding: black paint, green paint, yellow paint, white paint, magnetic paint, rubber, tar foam, soil/sand, water, brick, and terra cotta pot
Procedure:
•Place tested materials on corrugated cardboard to insulate from the counter tip
•Place 1ft2 of tested material 12cm below a 60w incandescent bulb
•Take the initial temperature of tested materials
•Turn on light bulb
•Take temperature every 5min for 20 min
•Turn off the light bulb
•Take temperature every 5min for 20min
•Find total temperature gain and lose for each material
•Compare materials and analyze data
For the first time, we made up a lab as a class. We had to design an experiment to test all of the possible materials we might use in the interior or exterior of our structure. First, we came up with a problem to solve. Which building materials are the most effective conductor/insulator/absorbing/sustaining heat? Then, we came up with materials list:
Conducting: Copper, plexiglas, aluminum, steel, plywood, OSB, screws, and nails
Insulating: double & single paned window, fiberglass insulation, magnifying glass, ridged foam, and spray foam.
Absorbing & Holding: black paint, green paint, yellow paint, white paint, magnetic paint, rubber, tar foam, soil/sand, water, brick, and terra cotta pot
Procedure:
•Place tested materials on corrugated cardboard to insulate from the counter tip
•Place 1ft2 of tested material 12cm below a 60w incandescent bulb
•Take the initial temperature of tested materials
•Turn on light bulb
•Take temperature every 5min for 20 min
•Turn off the light bulb
•Take temperature every 5min for 20min
•Find total temperature gain and lose for each material
•Compare materials and analyze data
7. Building Design
For our final project, we had to use all the knowledge from the previous labs to design a solar reflector panel and a cold frame. We had to design a scale model, materials list, blue prints, budget, key features, and presentation. For the scale model, we used 123D design to make our model so we didn't need to make separate blue prints for our cold frame and reflector. On March 11th 2015 we presented our two designs to our community.
For our final project, we had to use all the knowledge from the previous labs to design a solar reflector panel and a cold frame. We had to design a scale model, materials list, blue prints, budget, key features, and presentation. For the scale model, we used 123D design to make our model so we didn't need to make separate blue prints for our cold frame and reflector. On March 11th 2015 we presented our two designs to our community.
8. Justification
Many people think why would we want to create an daylight efficient home? As a class, we made a chart to show how much energy humans use and ways of generating electricity pros and cons for all, economic cost of electricity, the greenhouse effect and climate change.
Many people think why would we want to create an daylight efficient home? As a class, we made a chart to show how much energy humans use and ways of generating electricity pros and cons for all, economic cost of electricity, the greenhouse effect and climate change.
9. Generation of Electricity
The final part of our project was to design a turbine powered by wind to generate the electricity. We had to design a Vertical axis wind turbine (VAWT) and a horizontal axis wind turbine (HAWT). From our experiment, we learned that HAWT work more efficiently than VAWT.
The final part of our project was to design a turbine powered by wind to generate the electricity. We had to design a Vertical axis wind turbine (VAWT) and a horizontal axis wind turbine (HAWT). From our experiment, we learned that HAWT work more efficiently than VAWT.
Reflection:
This project was the longest project we've done so far. It was fun but at the same time it was very hard. From this project, I definetly learned many things. My group and I, we started this project really well. Everyone worked together to complete the task. However, later on, we kind of fell apart. For the solar water heater, everyone equally contributed. As the time went on, we slowly fell apart and Spencer and I ended up doing majority of the work. This was one of our pit. Another pit would be getting off topic. People in our group were very social that majority of us went off topic a lot. We talked about everything else BUT about our project. This led us to confusion. At one point, we all didn't know what we were doing. However, we had time to grade each other on how we are doing as a group. This was when we got to really talk about our problem in our group and solve it.
One of the pits would be time management. Even though we were off topic many times, we found our problem quick enought that we were able to finish our project in time. We had two days to practice for our presentation. Another thing I was able to improve on was being patient. There were times when one of my group member didn't want to contribute and didn't want to talk to us. At first, I was very frustrated because this is a group work and I wanted everyone to be equally contributed but he didn't want to. However, I was able to learn to be patient and take care of other's feelings. As the time went on, I was able to understand him more. Overall, there many problems within our group, but each one us learned many things from this project and we completed this project very successfully.
This project was the longest project we've done so far. It was fun but at the same time it was very hard. From this project, I definetly learned many things. My group and I, we started this project really well. Everyone worked together to complete the task. However, later on, we kind of fell apart. For the solar water heater, everyone equally contributed. As the time went on, we slowly fell apart and Spencer and I ended up doing majority of the work. This was one of our pit. Another pit would be getting off topic. People in our group were very social that majority of us went off topic a lot. We talked about everything else BUT about our project. This led us to confusion. At one point, we all didn't know what we were doing. However, we had time to grade each other on how we are doing as a group. This was when we got to really talk about our problem in our group and solve it.
One of the pits would be time management. Even though we were off topic many times, we found our problem quick enought that we were able to finish our project in time. We had two days to practice for our presentation. Another thing I was able to improve on was being patient. There were times when one of my group member didn't want to contribute and didn't want to talk to us. At first, I was very frustrated because this is a group work and I wanted everyone to be equally contributed but he didn't want to. However, I was able to learn to be patient and take care of other's feelings. As the time went on, I was able to understand him more. Overall, there many problems within our group, but each one us learned many things from this project and we completed this project very successfully.